By Douglas Starr, Jan. 24, 2019
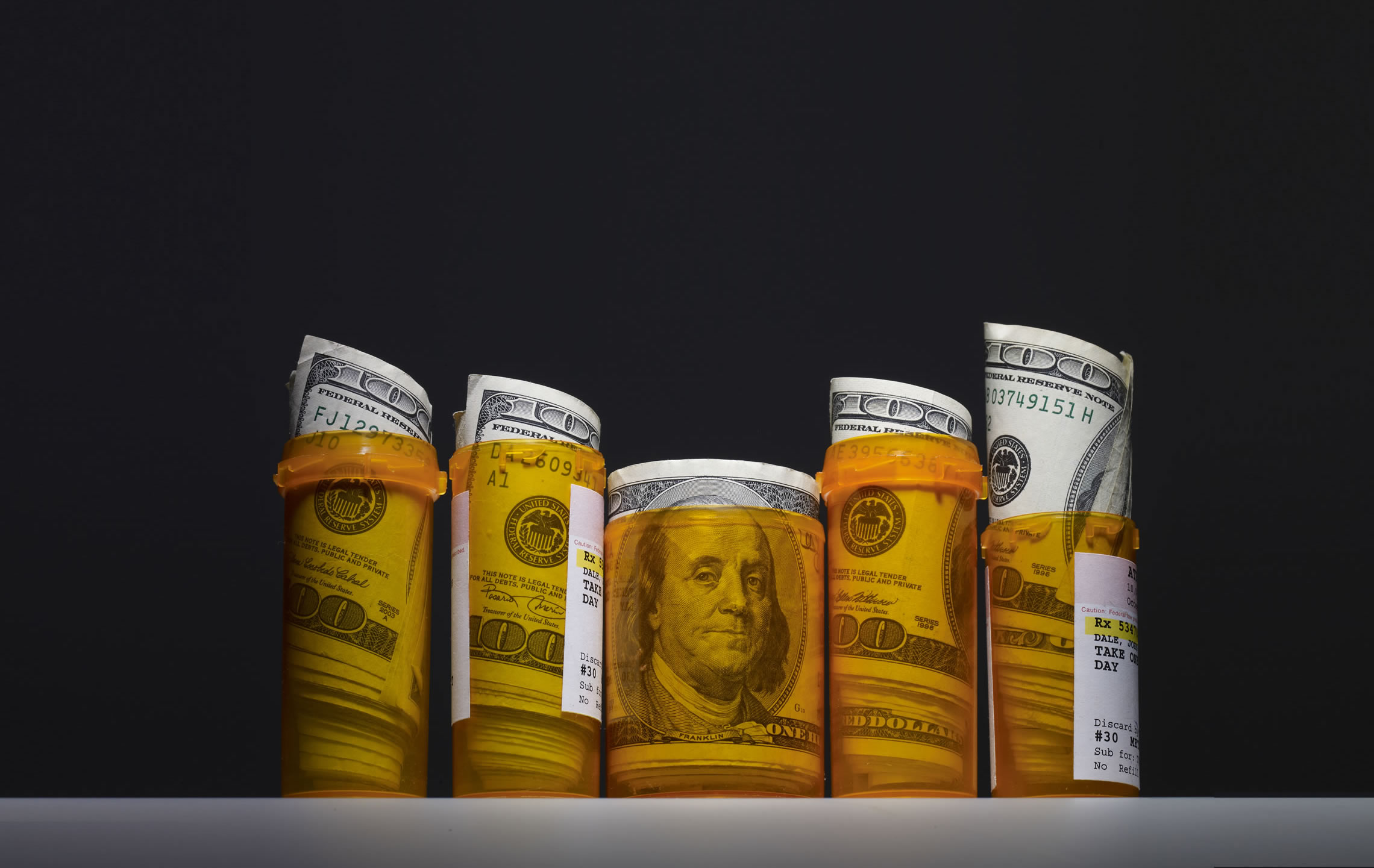
In a St. Louis, Missouri, courtroom last summer, David Egilman testified in a lawsuit filed by 22 women who claimed to have contracted ovarian cancer from exposure to Johnson & Johnson (J&J) baby powder. Like millions of women before them, they had dusted their babies with the powder and used it on themselves thousands of times. They alleged the talc was tainted with asbestos and that exposure to the carcinogenic fiber likely played a role in their cancers.
Egilman, a professor of family medicine at Brown University who served as a paid expert witness, brought science and medical gravitas to his testimony. He interviewed the women about their frequency and duration of talc use, and he factored in the levels of asbestos that outside scientists had found in samples of talc, which is sometimes mined from formations that also yield asbestos. From those data, he said, he could calculate the women’s doses of the carcinogen, and he argued that their exposure had doubled their risk of ovarian cancer.
J&J had its own experts, who said the talc was asbestos-free and there was no proof that the product caused cancer. But Egilman’s decisive contribution may have come from his scrutiny of company documents. After examining thousands of pages of internal J&J documents unearthed during the litigation, he and his student researchers concluded that J&J found no asbestos in the talc because its tests were not sensitive enough. He compared the company’s methods to trying to weigh a needle on a bathroom scale.”His bathroom scale analogy was brilliant,” says Mark Lanier, the Houston, Texas–based attorney who represented the women. “I actually put needles on a scale in front of the jury, several times.”
After a 6-week trial, the jury awarded $4.69 billion to the 16 surviving women and the families of the six who had died. It was the largest award in Missouri’s history, and it may foreshadow thousands of pending cases. J&J has appealed the decision. (Since the trial, other internal J&J documents have become public, which show the company knew about the asbestos contamination for decades but suppressed the information.)
Egilman believes corporations have minimized their costs at the expense of their employees’ health and that of the public and the environment—and that recent weakening of regulations has made that problem worse. He sees speaking out in court as a mission. “As a doctor, I can treat one cancer patient at a time,” he explained during a trial last year. “But by being here, I have the potential to save millions.” More broadly, he says corporate money and power have intimidated scientists and corrupted science itself—a concern that has led him into battle not only with corporations, but also with journals that publish what he describes as tainted results.
In 35 years as an expert witness, he has given depositions and testimony in more than 600 cases of occupational or environmental disease. He has helped win billions of dollars for injured or sick workers or consumers, or for the families of those who have died. By his reckoning, he has earned more than $5 million for such legal work; he says he has donated some of his fees to charities, including a nonprofit he founded to improve health in developing countries.
Specializing in occupational lung disease, Egilman diagnoses patients and marshals data. But he also digs into corporate records uncovered during litigation, invariably finding memos and studies showing that companies knew about industrial hazards long before warning employees or the public.
“He’s a bloodhound who can sniff out corporate misconduct better than security dogs at an airport,” Lanier says. Egilman’s opponents say he stretches science to the breaking point in his zeal to fight corporate malfeasance. “I think he’s a dangerous person,” said Patrick Hessel, an epidemiological consultant at EpiLung Consulting in Spruce Grove, Canada, who has tussled with Egilman over studies of auto mechanics’ exposure to asbestos. But scientists mostly defend him.
“He’s very strident about his views and pushes the public health and political side of things. … But I’m not aware of any wrong science,” says Arthur Frank, an environmental and occupational health researcher at Drexel University in Philadelphia, Pennsylvania. Frank says the link between asbestos-contaminated talc and cancer, for example, is well-established. “If you check his literature, he will document what he believes.”
“He is outspoken, unorthodox, brilliant—and driven to right wrongs,” says Harlan Krumholz, a Yale School of Medicine cardiologist who worked with Egilman in exposing the missteps of Merck & Co. in marketing the anti-inflammatory drug rofecoxib (Vioxx). “He is outside the mainstream academia but is an extraordinarily influential academic physician. He is a force, in style and substance.”
Courtroom activism
Stocky and bald, Egilman combines the passion of Ralph Nader with the timing of a Borscht Belt comedian. If you ask him a question, you’re likely to get a 20-minute response peppered with quips and rhetorical questions, along with hundreds of pages of supporting documents. Outside the courtroom, he sprinkles his answers with obscenities, referring mostly to the corporations he opposes. “I’m in the credit-giving business,” he says. “I believe that companies deserve full credit for lying, cheating, and endangering people’s health.”
The small research firm that serves as his base is called Never Again Consulting. The name refers to both the Holocaust (his father was survivor) and his research on corporate “screwups” so they never happen again. The firm occupies offices in Attleboro, Massachusetts, about 20 minutes from Brown. On any given day, the offices are abuzz with students from the university and local high schools, whom he hires for his research.
Last summer, four of his student helpers were developing a national education program about chronic traumatic encephalopathy, a degenerative brain disease linked to concussions that affects football players and other contact sport athletes. The students were also helping Egilman prepare to give a recorded deposition in the case of Greg Ploetz, a former University of Texas football player whose widow was suing the National Collegiate Athletic Association (NCAA) for his death. Backed by his students’ research, Egilman argued NCAA had known of the dangers of concussion for decades and failed to warn or safeguard its players. NCAA settled with Ploetz’s widow for $2 million.
“You see?” Egilman says. “I’ve been beating the biggest organizations in the world with 20-year-old kids.”
He grew up with a natural sympathy for the underdog. Born in 1952, Egilman was raised in a working class suburb of Boston. His father, Felix, a cobbler in his native Poland, attributed his survival in the Buchenwald concentration camp to a German officer who appreciated his shoemaking skills. After the war, having lost everything, including a wife and two sons, Felix came to the United States to make a new life. He remarried, got a job at the StrideRite shoe factory in Boston, bought a small house, and had a son, David.
But tragedy would pursue them. When David Egilman was 10 years old, his mother was killed in a car accident. His father never spoke much; Egilman, who did all the shopping and housework, had to find fuel for his ambitions outside the home. Fortunately, some excellent elementary and junior high school teachers took him under their wing. “That’s where I got my nurturing, more or less,” he once told a reporter.
Encouraged by his teachers to go to college, Egilman won scholarships to Brown, where he earned his bachelor’s and medical degrees and volunteered for VISTA, a national antipoverty program. After earning a master’s degree in public health from Harvard University, he moved to Cincinnati, Ohio, where he worked for the National Institute for Occupational Safety and Health (NIOSH). He also started a workers’ clinic funded by AFL-CIO and did pro bono consulting for a union whose members worked in a nuclear weapons plant.
Products on Trial
In more than 30 years of courtroom appearances, public health expert David Egilman has testified against products—and their makers—that he believes put the welfare of workers or the public at risk. Here are some of his highest-profile targets.
In 1984, he took his activism into the courtroom. While at NIOSH, he had published a health hazard evaluation on isocyanate, an industrial chemical that killed thousands of people after an explosion at a Union Carbide pesticide plant in Bhopal, India. An attorney contacted him to testify for a spray painter at an auto body shop who had suffered lung damage from isocyanate, which is also a component of automotive spray paint. Egilman testified, and the company settled. “It turned out I was a natural,” he says.
Sometime later, he was giving a talk about the case when he was approached by an attorney representing a pipe fitter who had been exposed to asbestos. Egilman testified in court that the man showed irreversible lung damage and had a greater than 50% chance of dying from asbestos-related disease. (Witnesses representing the employer disagreed, but 20 years later, the man died of mesothelioma, a cancer caused almost exclusively by asbestos.) The jury ordered the company to pay $26.3 million, an amount eventually reduced to $1.5 million.
Word spread that Egilman was an authoritative witness whose common touch made him effective with juries. He began to accept more and more cases—15 or more per year. His opponents see his caseload as evidence that he is far from a dispassionate expert. “Anything that he can testify to, he will,” says Tom Radcliffe, a partner with the Baltimore, Maryland–based law firm DeHay & Elliston who has gone up against Egilman.
“I don’t deny being an advocate for public health,” Egilman says. “But my opinions cannot extend beyond the science. These are the largest corporations in the world, with unlimited resources. If I get too far out over my skis, they’ll cut my legs off.”
He left NIOSH and returned to Brown, where he teaches just one course, Science and Power: The Corruption of Public Health. It leaves him time to do research in global public health and take part in litigation. The course also serves as a fertile ground for recruiting the students who help him dig through company files and find the incriminating memos that often win cases.
In 2005, Egilman was an expert witness in the first major tort case against Merck, the maker of Vioxx. He gained access to thousands of company memos showing Merck did not pursue early study data indicating possible cardiovascular risks and hid those data from the Food and Drug Administration (FDA). Not until 2002 did Merck add warning labels to the drug, at FDA’s request; and not until 2004 did the company voluntarily withdraw Vioxx from the market after further studies showed serious cardiovascular risks. Meanwhile, 107 million Vioxx prescriptions had been filled, resulting in tens of thousands of heart attacks.
Egilman testified on behalf of the widow and daughter of a 56-year-old man who died of a heart attack after taking Vioxx. The jury awarded them $24.4 million and assessed an additional $253 million in punitive damages. Two years later, Merck agreed to pay $4.85 billion to settle the remaining 27,000 lawsuits, according to press reports.
Reviewing company memos, Egilman found evidence of other questionable practices. One was “ghostwriting,” in which Merck’s marketing department would write a rough draft of a paper, recruit an outside scientist to edit or contribute to it, and then list the hired scientist as the primary author. “I found an email saying, ‘Who should we have as an author on this paper?’” Egilman recalls, “and another listing the author as ‘Dr. to be determined.’” When he and colleagues published those findings in JAMA in 2008, the journal’s editors wrote a commentary calling ghostwriting “unprofessional and demeaning to the medical profession and to scientific research.”
The Merck documents also revealed evidence of “seeding,” in which a marketing department designs clinical studies whose real purpose is to promote a drug before or during FDA review. The goal is to create a ready market on approval. In this case, Merck recruited 600 doctors to conduct a trial designed “to accelerate the uptake of Vioxx,” according to an internal memo. “It may be a seeding study,” another memo said, “but let’s not call it that.”
Egilman and several colleagues, including Krumholz, described their findings in the Annals of Internal Medicine, whose editors denounced the practice as “marketing in the guise of science.” In a rebuttal letter to the journal, a Merck scientist said the study was a “double-blind, randomized, controlled clinical trial with a legitimate scientific purpose” in testing how well patients tolerated Vioxx.
Pressing Boundaries
Egilman’s sense of mission propels a prodigious work ethic—by his own estimate, he works 100 to 120 hours per week—and sometimes emboldens him to act rashly. In 2007, patients were suing Eli Lilly and Company, claiming that its antipsychotic medicine olanzapine (Zyprexa) had caused dramatic weight gain and diabetes. Reviewing company documents as an expert witness for the plaintiffs, Egilman found emails implying Eli Lilly knew of the danger but long tried to play it down. The judge ordered that the documents be kept confidential to protect the company’s proprietary sales and marketing strategies, but Egilman leaked them to a New York Times reporter. “A physician’s oath never says to keep your mouth shut,” Egilman says.
The court was not amused by that “brazen breach” of protocol. “They could have put me in jail,” Egilman says. To avoid criminal charges, he paid a $100,000 settlement. But after the newspaper story ran, 30 states subpoenaed documents on Eli Lilly’s sales and marketing activities, showing the same incriminating behavior. In 2009, it agreed to pay $1.4 billion to settle criminal charges and civil suits.
Egilman presses boundaries behind the scenes as well. Like other expert witnesses, he gives depositions before trial as part of the fact-finding process. Some attorneys use depositions for another purpose—to wear down witnesses or even discourage them from testifying by asking scores of detailed or accusatory questions.
“You have no idea how searching and oppressive it can be,” says Barry Castleman, a nationally known asbestos expert in Garrett Park, Maryland, who says he has been deposed more than 1000 times. “They take everything about your past, everything you’ve ever written or done, everything you’ve ever said and throw it in your face, looking for errors, ambiguities, inconsistencies.”
“It’s basically a control issue,” Egilman says. His response is to harass or unnerve opponents. On occasion, he has shown up to a deposition in fuzzy slippers and a dashiki, over which he wore a physician’s white jacket and his Harvard tie. Sometimes he answers simple questions in exquisite and unnecessary detail, dragging out the process for hours. Often he’ll use the conference room as a stage set, setting up posters and bookshelves to visually reinforce what he plans to say—and to discomfit opposing attorneys.
In 2002, a lawyer for Brush Wellman Inc.—a supplier of beryllium to the aerospace and electronics industries that workers were suing for beryllium-related lung disease—formally complained about Egilman’s behavior. Arriving to take Egilman’s deposition, the lawyer found that the conference room had been set up with shelves full of books, including many with provocative titles about Nazi Germany. The witness table was covered with Egilman’s papers and notebooks, one labeled “Brush Wellman’s Lies.”
The attorney picked up one of Egilman’s books and began to peruse it. The next thing he knew, Egilman was inches from his face, shouting at him to put the book down. “I felt as though he was going to physically attack me,” the attorney wrote, asking that Egilman be excluded from testifying “and perhaps locked up.” Both requests were denied.
At Odds with a Publisher
Last year, Egilman found himself the focus of a dispute that he says reveals another dimension of corporate influence over science—the attempt to censor scientific literature. In 2015, a giant of the science publishing world, Taylor & Francis, bought the International Journal of Occupational and Environmental Health, which Egilman had edited for the previous decade. The journal, with its small but devoted readership, had a reputation for opposing “mercenary science”—the hiring of scientists to produce industry-friendly studies—according to epidemiologist David Michaels, a former U.S. assistant secretary of labor for occupational safety and health who is now at George Washington University in Washington, D.C.
The new publisher let Egilman’s contract expire and in 2017 withdrew a peer-reviewed paper of his that the journal had already published. That paper harshly criticized an earlier study claiming to show worker exposure to asbestos was lower than previously assumed during the manufacture of Bakelite, a kind of plastic. In his paper, Egilman charged that the study was nothing more than “data manipulation masquerading as science” to defend industry.
Taylor & Francis gave no reason for withdrawing Egilman’s paper or for appointing a new editor, who drew criticism because he had consulted for industry. In response, the entire editorial board resigned. Thirty current and past board members and the founding editor-in-chief then took the unusual step of asking the National Library of Medicine to rescind the journal’s listing in MEDLINE, expressing concern about the “ascendancy of corporate interests over independent science in the public interest.”
The publisher told the blog Retraction Watch that Egilman’s article had been “published inadvertently, before the review process was completed,” and was later deemed “unsuitable for publication.” The journal remains listed, though it has announced plans to cease publication.
Not Intimidated
Egilman was under fire again last spring. He had published a paper in the journal Accountability in Research, alleging J&J had committed “grave fraudulence” in a study of hip prostheses produced by one of its subsidiaries. The device, a metal-on-metal hip replacement, was taken off the market in 2013 after thousands of people suffered tissue damage and metal poisoning. The company has been ordered to pay nearly $250 million in damages and still faces lawsuits from thousands of patients. Egilman, who gained access to internal documents, charged that all that pain and suffering was unnecessary—that the company’s clinical trial of the device was no more than a seeding study and that J&J had covered up the device’s shortcomings.
A biostatistician from J&J wrote a six-page letter to the journal hinting at legal action and demanding that the editors retract Egilman’s article. The letter charged that Egilman had committed many factual errors and failed to disclose his conflict of interest as an expert witness for the patients. Not to be intimidated, Egilman released the letter to the press, along with portions of his 32-page rebuttal. He called the letter yet another attempt “to bully the scientific and medical community.” His paper remains in print.
Most people would find such conflicts stressful, but they energize Egilman. “As [a] physician, I took an oath that included a responsibility to public health, not just to individual care,” he says. Fighting powerful interests can mean “being viewed as a weirdo, an outsider. That’s the downside, but one that you have to be willing to accept.”